Description
Deleobuvir, also known as BI207127, a non-nucleoside hepatitis C virus NS5B polymerase inhibitor for the treatment of hepatitis C. Deleobuvir was tested in combination regimens with pegylated interferon and ribavirin, and in interferon-free regimens with other direct-acting antiviral agents including faldaprevir. Deleobuvir showed that a triple combination of deleobuvir, faldaprevir, and ribavirin performed well in HCV genotype 1b patients. Efficacy fell below 50%, however, for dual regimens without ribavirin and for genotype 1a patients. In December 2013, deleobuvir was discontinued since recent findings from phase III trials did not suggest sufficient efficacy.
Product information
CAS Number: 863884-77-9
Molecular Weight: 653.57
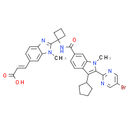
Formula: C34H33BrN6O3
Synonym:
BI207127
BI-207127
Deleobuvir
Smiles: CN1C(=NC2=CC=C(C=C12)/C=C/C(O)=O)C1(CCC1)NC(=O)C1=CC2=C(C=C1)C(C1CCCC1)=C(C1N=CC(Br)=CN=1)N2C
InChiKey: BMAIGAHXAJEULY-UKTHLTGXSA-N
InChi: InChI=1S/C34H33BrN6O3/c1-40-26-17-22(10-11-24(26)29(21-6-3-4-7-21)30(40)31-36-18-23(35)19-37-31)32(44)39-34(14-5-15-34)33-38-25-12-8-20(9-13-28(42)43)16-27(25)41(33)2/h8-13,16-19,21H,3-7,14-15H2,1-2H3,(H,39,44)(H,42,43)/b13-9+
Technical Data
Appearance: Solid Power
Purity: ≥98% (or refer to the Certificate of Analysis)
Solubility: To be determined
Shipping Condition: Shipped under ambient temperature as non-hazardous chemical or refer to Certificate of Analysis
Storage Condition: Dry, dark and -20 oC for 1 year or refer to the Certificate of Analysis.
Shelf Life: ≥12 months if stored properly.
Stock Solution Storage: 0 - 4 oC for 1 month or refer to the Certificate of Analysis.
Drug Formulation: To be determined
HS Tariff Code: 382200
References:
- Sane RS, Ramsden D, Sabo JP, Cooper C, Rowland L, Ting N, Whitcher-Johnstone A, Tweedie DJ. Contribution of major metabolites towards complex drug-drug interactions of deleobuvir: in vitro predictions and in vivo outcomes. Drug Metab Dispos. 2015 Dec 18. pii: dmd.115.066985. [Epub ahead of print] PubMed PMID: 26684498.
- Asselah T, Zeuzem S, Soriano V, Bronowicki JP, Lohse AW, Müllhaupt B, Schuchmann M, Bourlière M, Buti M, Roberts SK, Gane EJ, Stern JO, Voss F, Baum P, Gallivan JP, Böcher WO, Mensa FJ. ITPA Genotypes Predict Anemia but Do Not Affect Virological Response with Interferon-Free Faldaprevir, Deleobuvir, and Ribavirin for HCV Infection. PLoS One. 2015 Dec 9;10(12):e0144004. doi: 10.1371/journal.pone.0144004. eCollection 2015. PubMed PMID: 26650626; PubMed Central PMCID: PMC4674133.
- McCabe M, Sane RS, Keith-Luzzi M, Xu J, King I, Whitcher-Johnstone A, Johnstone N, Tweedie DJ, Li Y. Defining the Role of Gut Bacteria in the Metabolism of Deleobuvir: In Vitro and In Vivo Studies. Drug Metab Dispos. 2015 Oct;43(10):1612-8. doi: 10.1124/dmd.115.064477. Epub 2015 Jun 11. PubMed PMID: 26068924.
- Yatsuhashi H, Kodani N, Ugai H, Omata M. Open-label phase 2 study of faldaprevir, deleobuvir and ribavirin in Japanese treatment-naive patients with chronic hepatitis C virus genotype 1 infection. Hepatol Res. 2015 May 20. doi: 10.1111/hepr.12535. [Epub ahead of print] PubMed PMID: 25991083.
- Latli B, Hrapchak M, Chevliakov M, Li G, Campbell S, Busacca CA, Senanayake CH. Synthesis of deleobuvir, a potent hepatitis C virus polymerase inhibitor, and its major metabolites labeled with carbon-13 and carbon-14. J Labelled Comp Radiopharm. 2015 May 30;58(6):250-60. doi: 10.1002/jlcr.3294. Epub 2015 May 11. PubMed PMID: 25964148.
Products are for research use only. Not for human use.
Payment & Security
Your payment information is processed securely. We do not store credit card details nor have access to your credit card information.