Description
GTS-21 2HCl, also known as DMBX-A, is a derivative of the natural product anabaseine that acts as a partial agonist at neural nicotinic acetylcholine receptors. It binds to both the α4β2 and α7 subtypes, but activates only the α7 to any significant extent. Both GTS-21 itself and its demethylated active metabolite 4-OH-GTS-21 display nootropic and neuroprotective effects, and GTS-21 is being investigated for the treatment of Alzheimer's disease, nicotine dependence, and, most significantly, for schizophrenia.
Product information
CAS Number: 156223-05-1
Molecular Weight: 381.30
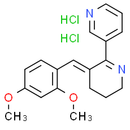
Formula: C19H22Cl2N2O2
Synonym:
GTS-21
GTS-21 hydrochloride
GTS-21 HCl
DMBX-A
DMBX-anabaseine
GTS-21 dihydrochloride
GTS 21
GTS21
Related CAS Number:
148372-04-7 (free base)
Chemical Name: 3-[(3E)-3-[(2,4-dimethoxyphenyl)methylidene]-5,6-dihydro-4H-pyridin-2-yl]pyridine dihydrochloride
Smiles: Cl.Cl.COC1C=C(OC)C(/C=C2\CCCN=C\2C2=CN=CC=C2)=CC=1
InChiKey: BXKYFUGAAFLYJL-BXGYHSFXSA-N
InChi: InChI=1S/C19H20N2O2.2ClH/c1-22-17-8-7-14(18(12-17)23-2)11-15-5-4-10-21-19(15)16-6-3-9-20-13-16;;/h3,6-9,11-13H,4-5,10H2,1-2H3;2*1H/b15-11+;;
Technical Data
Appearance: Solid Power
Purity: ≥98% (or refer to the Certificate of Analysis)
Solubility: Soluble in DMSO, not in water
Shipping Condition: Shipped under ambient temperature as non-hazardous chemical or refer to Certificate of Analysis
Storage Condition: Dry, dark and -20 oC for 1 year or refer to the Certificate of Analysis.
Shelf Life: ≥12 months if stored properly.
Stock Solution Storage: 0 - 4 oC for 1 month or refer to the Certificate of Analysis.
Drug Formulation: To be determined.
HS Tariff Code: 382200
How to use
In Vitro:
GTS-21 bound to human α4β2 nAChR (Ki=20 nM) 100-fold more potently than to humanα7-nAChR, and is 18- and 2-fold less potent than (-)-nicotine at human α4β2 and a7 nAChR, respectively.
In Vivo:
GTS 21 (4 mg/kg; i.p.; 1, 3, 7, 14 and 21 days) reduces radiation induced histological signs of pulmonary injury.
References:
- Yue Y, Liu R, Cheng W, Hu Y, Li J, Pan X, Peng J, Zhang P. GTS-21 attenuates lipopolysaccharide-induced inflammatory cytokine production in vitro by modulating the Akt and NF-κB signaling pathway through the α7 nicotinic acetylcholine receptor. Int Immunopharmacol. 2015 Oct 17. pii: S1567-5769(15)30138-7. doi: 10.1016/j.intimp.2015.10.005. [Epub ahead of print] PubMed PMID: 26490221.
- Kong FJ, Ma LL, Zhang HH, Zhou JQ. Alpha 7 nicotinic acetylcholine receptor agonist GTS-21 mitigates isoflurane-induced cognitive impairment in aged rats. J Surg Res. 2015 Mar;194(1):255-61. doi: 10.1016/j.jss.2014.09.043. Epub 2014 Oct 5. PubMed PMID: 25450597.
- Wu S, Zhao H, Luo H, Xiao X, Zhang H, Li T, Zuo X. GTS-21, an α7-nicotinic acetylcholine receptor agonist, modulates Th1 differentiation in CD4(+) T cells from patients with rheumatoid arthritis. Exp Ther Med. 2014 Aug;8(2):557-562. Epub 2014 Jun 3. PubMed PMID: 25009619; PubMed Central PMCID: PMC4079428.
- Hu Y, Liu R, Li J, Yue Y, Cheng W, Zhang P. Attenuation of Collagen-Induced Arthritis in rat by nicotinic alpha7 receptor partial agonist GTS-21. Biomed Res Int. 2014;2014:325875. doi: 10.1155/2014/325875. Epub 2014 Feb 27. PubMed PMID: 24719855; PubMed Central PMCID: PMC3955649.
- Sitapara RA, Antoine DJ, Sharma L, Patel VS, Ashby CR Jr, Gorasiya S, Yang H, Zur M, Mantell LL. The α7 nicotinic acetylcholine receptor agonist GTS-21 improves bacterial clearance in mice by restoring hyperoxia-compromised macrophage function. Mol Med. 2014 Jun 19;20:238-47. doi: 10.2119/molmed.2013.00086. PubMed PMID: 24664237; PubMed Central PMCID: PMC4069272.
- Briggs CA, et al. Functional characterization of the novel neuronal nicotinic acetylcholine receptor ligand GTS-21 in vitro and in vivo. Pharmacol Biochem Behav. 1997;57(1-2):231-241.
- Zhang R, et al. N-terminal domains in mouse and human 5-hydroxytryptamine3A receptors confer partial agonist and antagonist properties to benzylidene analogs of anabaseine. J Pharmacol Exp Ther. 2006;317(3):1276-1284.
- Mei Z, et al. α7 nAchR agonist GTS 21 reduces radiation induced lung injury. Oncol Rep. 2018;40(4):2287-2297.
Products are for research use only. Not for human use.
Payment & Security
Your payment information is processed securely. We do not store credit card details nor have access to your credit card information.